By Emily Leclerc, Waisman Science Writer
Fragile X syndrome (FXS) is the most common heritable form of intellectual and developmental disability. It is also the most common single genetic contributor to autism spectrum disorder (ASD). But like all intellectual and developmental disorders, FXS and ASD are molecularly complex and researchers are still endeavoring to understand them at the cellular and molecular level. A new paper by Waisman investigator Xinyu Zhao, PhD, professor of neuroscience and Jenni and Kyle Professor in Novel Neurodevelopmental Diseases, illuminates another molecular feature that contributes to issues seen in FXS and ASD.
The study, which was published in the journal Nature Communications, showcases the relationship between elevated levels of the protein MAP1B and ASD and FXS symptoms and a potential treatment using the FDA-approved drug rapamycin that rescues the damage caused by high levels of MAP1B. MAP1B is a crucially important protein in developing neurons as it is an essential component of cells’ cytoskeleton. The loss of MAP1B is intolerable to humans. The push to investigate this protein was brought to Zhao, by a Waisman clinician.
Several years ago, a Waisman clinician approached Zhao to talk about one of their patients. The patient presented with severe autism and FXS-like symptoms and had an interesting mutation that had added multiple copies to a small region of a chromosome. “The clinician did genetic testing and didn’t find the mutation that causes fragile X,” Zhao says. “But they did find that the multiplication involved the gene for the protein MAP1B. When the clinician told us about this, the first thing we thought was that MAP1B might be a problem.”
To investigate this, Zhao, the first author Yu Guo, PhD, research scientist, and her lab created mouse models and human cell models with elevated MAP1B and found that it was detrimental. The mice presented with severe ASD-like behavioral patterns. She then decided to look further to see if these findings may be related to FXS.
It has been known for decades that the protein FMRP, which is dysregulated in FXS, is a regulator of MAP1B. A mutation in FMRP’s gene, FMR1, causes deficiencies in FMRP and is the cause of fragile X syndrome. Zhao thought that the FMRP deficiency could lead to elevated levels of MAP1B which might contribute to the neuron phenotypes seen in FXS. “We used two stem cell lines derived from individuals with fragile X published by my colleague Anita Bhattacharyya, [PhD, Waisman investigator and associate professor of cell and regenerative biology], and differentiated them into neurons and found they did have elevated MAP1B,” Zhao says.
So, elevated levels of MAP1B – whether by a gene multiplication or because of the FXS mutation – is harmful to neurons during development. Zhao then decided to look further to determine what exactly was happening to cells when MAP1B is elevated to determine why it is harmful. She found that the cell’s “self-destruction” or autophagy pathway was disrupted.
“Autophagy, for lack of better words, is self-eating. It is one of the ways proteins get turned over in the cell. It happens by either getting rid of proteins that you don’t need any more or they are damaged and you want to get rid of them,” Zhao says. This is an important process in cells as it controls how much waste is floating around. Too much detritus can impact the cell’s functioning.
MAP1B interacts with important components of the autophagy pathway. Zhao found that when the levels of MAP1B are elevated, it interacts too much with the autophagy pathway which prevents it from happening. “It is like MAP1B is trapping the cleaning crew and stopping them from cleaning up the city ,” Zhao says.
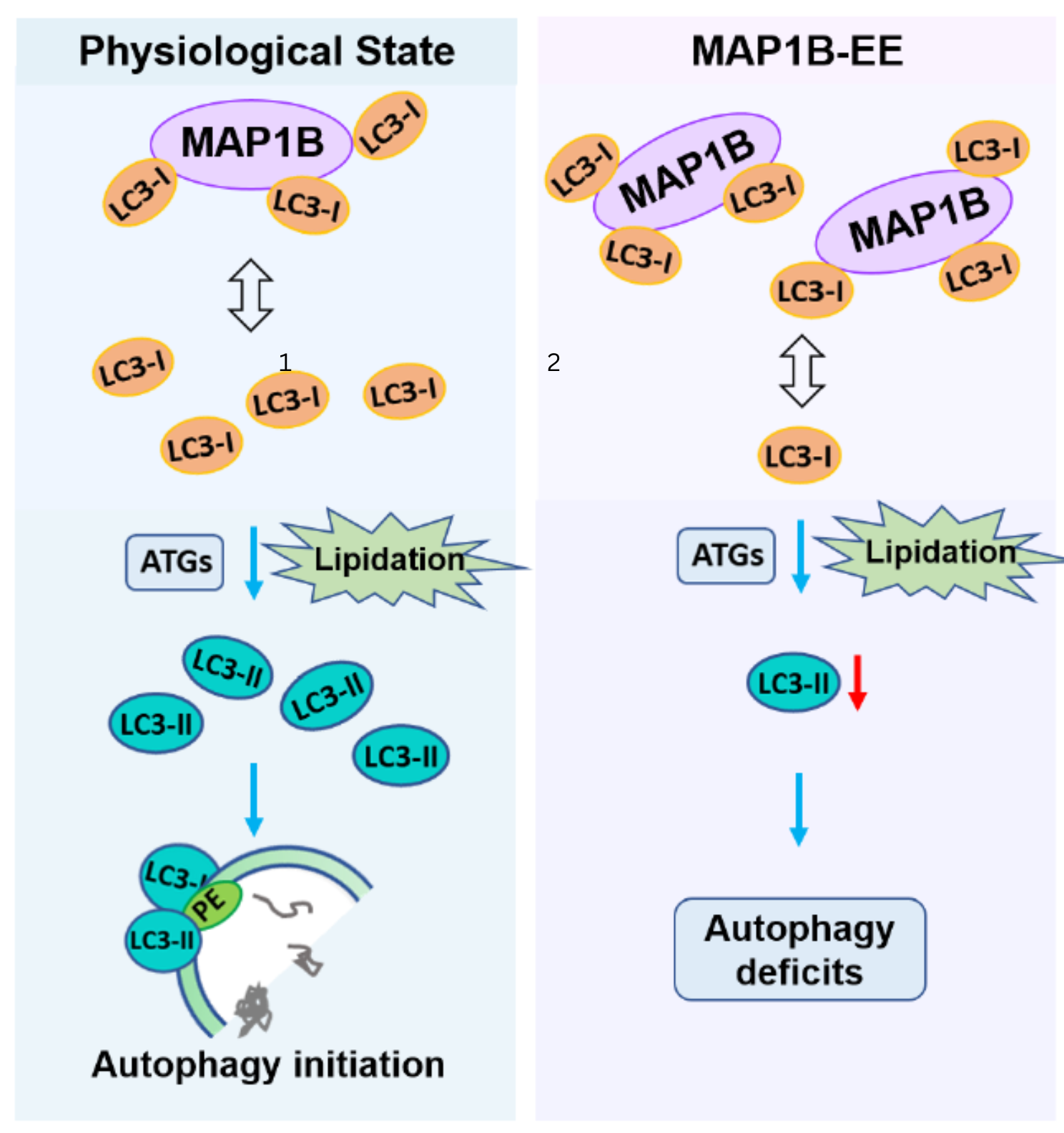
All of that work then led to the crux of the paper – can Zhao and her lab ameliorate the issues caused by MAP1B elevation by stimulating the autophagy pathway? They treated both ASD neurons and fragile X neurons with rapamycin, a drug known to activate autophagy. They found that the treated cells did have their function rescued. Then when they repeated that process in primate and human brain slices they altered in culture to mimic FXS, they again found that treating with rapamycin was able to correct the damage done to the neurons. This paper provides further evidence that rapamycin has an important impact on fragile X cells.
“We are not the first to consider rapamycin for fragile X because there was a mouse study in 2018 that showed that you can use rapamycin to correct some of fragile X’s phenotypes in mouse models,” Zhao says. “Our study is providing more proof and proof in human neuron models that it has an effect.” This paper also shows an important causal link between MAP1B elevation and deficits seen in both FXS and ASD.
Understanding the impacts of elevated MAP1B and that stimulating the autophagy pathway can rescue neuron function provides another potential treatment target for FXS and ASD. Zhao cautions though that there are still many steps between the current research and a possible treatment. Rapamycin may have many side effects and potential downsides. And while better understanding the effect of MAP1B elevation is another piece in the puzzle, there is a lot still unknown about FXS and ASD at a molecular level.
Zhao intends to continue this work by screening more drugs to see if others activate autophagy and by looking closer at FXS and ASD in neurons, human neurons, and organoids to better understand the other changes happening in the cells. Ultimately, she hopes her work could lead to a possible targeted gene therapy.
She also hopes this paper highlights the importance of clinicians and researchers working closely together. “The clinician saw the patient [that inspired this research] and realized the similarity between this autism patient and fragile X patients and actually paid attention,” Zhao says. “If the clinician hadn’t paid attention, we might have never known about this.” Waisman’s interdisciplinary environment of having clinics and research labs all housed in the same building helped to create the opportunity for this kind of interaction.
“If we can create more opportunities like this, more awareness by the clinicians and more interaction between the clinical side and the basic science side,” Zhao says, “we can make a lot of really novel discoveries.”

Several Waisman investigators contributed to the study including Qiang Chang, PhD, professor of medical genetics and neurology, Andre Sousa, PhD, assistant professor of neuroscience, and Daifeng Wang, PhD, associate professor of biostatistics and medical informatics, computer sciences. Zhao would also like to recognize MCP graduate student Natasha Mendez-Abello, and several UW undergraduate students Sabrina Huang, Jonathan Le, Ezra Jarembowski, Keegan Schoeller, Michael Stockton who contributed to this work.
This work was supported by grants from the National Institutes of Health (R01MH118827, R01NS105200, R01MH116582, R01HD064743, R01NS064025, R01AG067025, and U01MH116492, P51 OD011106 to the Wisconsin National Primate Research Center, U54HD090256 and P50HD105353 to the Waisman Center, and R24HD000836), DOD IIRA W81XWH2210621 grant, Brain Research Foundation, UW Vilas (Mid-Career Award), Wisconsin Alumni Research Foundation, Jenni and Kyle Professorship, National Science Foundation Career Award 2144475, NARSAD Young Investigator Grant from the Brain and Behavior Research Foundation 28721, T32 GM141013 Molecular Pharmacology training grant and SciMed scholarship, Simons SFARI pilot grant, FRAXA Postdoctoral fellowships, postdoctoral fellowships from UW Stem Cell and Regenerative Medicine Center and Autism Science Foundation, Hilldale Undergraduate Research Scholarships, and R24HD000836 (to Birth Defects Research Laboratory).
| Your support makes a difference. Donate now to advance knowledge about human development, developmental disabilities, and neurodegenerative diseases through research, services, training, and community outreach. | DONATE NOW |
 |
Learn more about the Waisman Center's 50th Anniversary, including events, history, stories and images: 50 Years | 1973 - 2023 |